Purpose: The purpose of this lab was to determine the optimum stoichiometric ratio between the reactants sodium hypochlorite (NaClO) and sodium thiosulfate (Na2S2O3) in a chemical reaction.
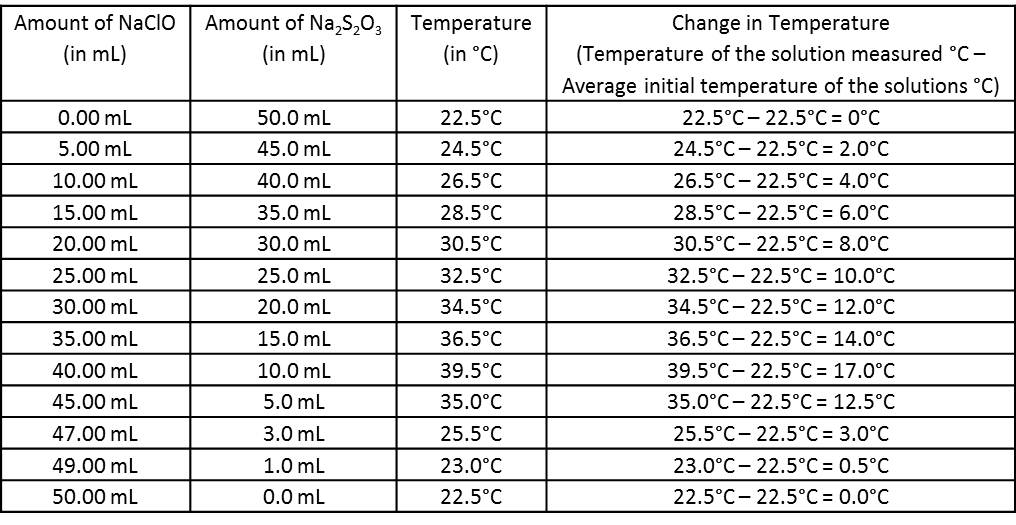
Data Table:
Data Table:
*The average initial temperature of the solutions was calculated before the lab began. It was the (temperature the NaClO solution + temperature of the Na2S2O3 solution)/2 = (22.5°C + 22.5°C)/2 = 22.5°C.
**For the NaClO solution, the 10mL graduated cylinder was used. Therefore, to account for uncertainty in measurement, the measurements were rounded to the hundredths place. For the Na2S2O3 solution, the 100mL graduated cylinder was used. Therefore, to account for uncertainty in measurements, the measurements were rounded to the tenths place.
***For the thermometer, the smallest unit of measurement was the ones place. Therefore, to account for uncertainty in measurement, the measurements were rounded to the tenths place.
Graph:
**For the NaClO solution, the 10mL graduated cylinder was used. Therefore, to account for uncertainty in measurement, the measurements were rounded to the hundredths place. For the Na2S2O3 solution, the 100mL graduated cylinder was used. Therefore, to account for uncertainty in measurements, the measurements were rounded to the tenths place.
***For the thermometer, the smallest unit of measurement was the ones place. Therefore, to account for uncertainty in measurement, the measurements were rounded to the tenths place.
Graph:
Conclusion: The optimum ratio (the stoichiometric ratio in the equation), in general, is the ratio of the solutions that cause the most reactants to be used up and the most products to be produced. For the chemical reaction between sodium hypochlorite (NaClO) and sodium thiosulfate (Na2S2O3) the amount of heat produced and the change in temperature were used to determine the optimum ratio between the two solutions.
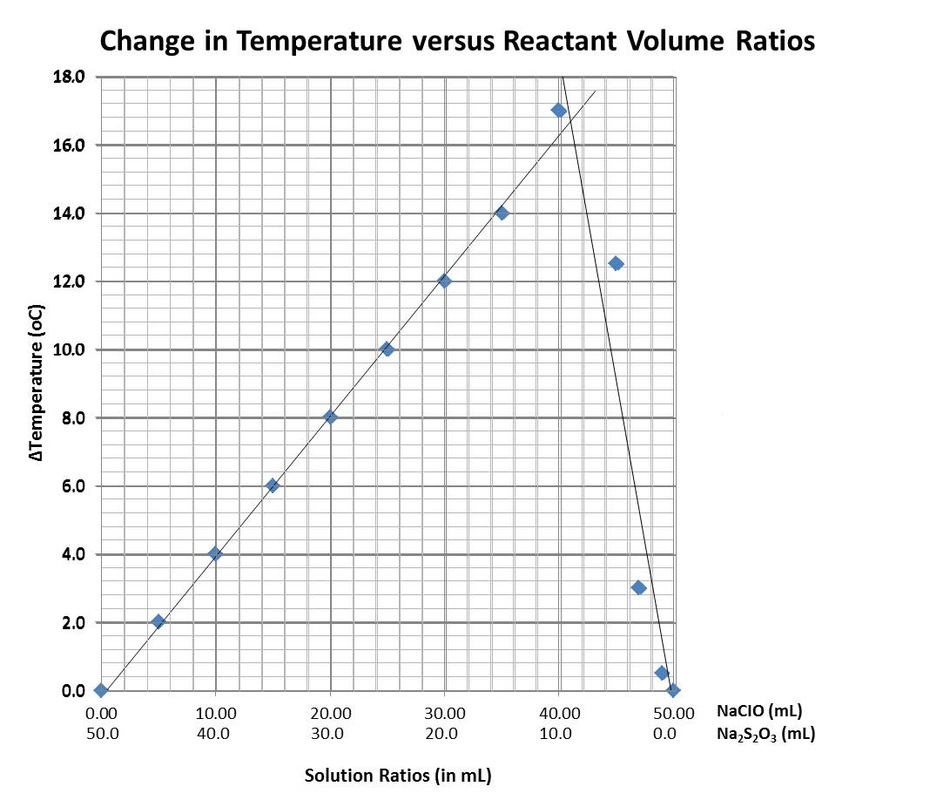
The ratio of the two solutions that produced the most heat and greatest temperature change was the optimum ratio. The optimum ratio of NaClO:Na2S2O3 in this chemical reaction was approximately 41:9 which equals 4.56:1, simplified. This ratio produced the highest temperature of 39.0°C and the greatest temperature change of 17.0°C.
Discussion of Theory: The coefficients of compounds in a balanced chemical equation represent the mole to mole ratio of the compounds in a chemical reaction. The ratio of the coefficients is known as the stoichiometric ratio and is the optimum ratio of the compounds to where the most reactants are used up and the most products are produced. When the products of a chemical reaction are unknown, it is not possible
to balance the equation. Therefore, experimental methods must be used to determine the stoichiometric ratio of the compounds. The method of continuous variations was used to determine the stoichiometric ratio between NaClO and N2S2O3 in this experiment. As part of this method, different ratios of the compounds NaClO and N2S2O3were mixed together. An exothermic reaction occurs between these two compounds, and this results in heat being released. By measuring the temperature of each mixture with the different ratios of the compounds, the optimum ratio was found. In this case, the mixture that released the most heat, which meant the most reactants were used up and the most products were produced, contains the optimum ratio of the two compounds. This optimum ratio is the stoichiometric ratio of the moles of the compounds.
Analysis Questions:
1. The differing amounts of NaClO and N2S2O3 combined to form mixtures served as the independent variable. In order for an experiment to yield true results, there can only be one independent variable, one thing that is changing. The volume served as a constant variable. The volume of the reactants needed to be kept constant because the ratios need to be made with the same volume of reactants. If volume was not kept constant, it would have been difficult to decide if the differing volumes were the result of a certain temperature reading or the ratio of the two compounds were the result of a certain temperature reading.
2. The limiting reagent is another way of saying the limiting reactant. The limiting reagent is the reactant in a chemical reaction that is completely used up and limits the amount of product that can be produced.
3. Volume limits the precision of the data in this experiment. Precision is how close a set of measurements are to each other. When different
ratios of NaClO and N2S2O3are mixed and the volume is kept constant, each ratio is going to produce a different amount of heat. There cannot be precision between different ratios of elements even if they have similar temperatures because they are different elements. In order for measurements to have precision, the measurements have to be of one ratio. If the volume were not kept constant, a ratio of 1:21 and 2:42 would yield similar temperature results because the first is the simplification of the second. Therefore, these measurements would have a high precision.
4. The limiting reagent along the upward sloping line of the graph is NaClO. The limiting reagent along the downward sloping line of the graph is the Na2S2O3.
5. Depending on the experiment, the method of continuous variations could use the mass of a precipitate formed, the volume of a gas released, or the changes in color intensity of a reactant or product.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio rather than the ratio associated with the greatest
temperature change because not all of the ratios of the compounds were tested. Although the highest temperature change resulted from the ratio of 40.0 mL NaClO and 10 mL Na2S2O3, a higher ratio could have resulted from a slightly higher or lower ratio. Since these were not tested and measured, using the line of best fit intersection gives a more accurate estimation of the ratio that yielded the highest temperature change.
The ratio of the two solutions that produced the most heat and greatest temperature change was the optimum ratio. The optimum ratio of NaClO:Na2S2O3 in this chemical reaction was approximately 41:9 which equals 4.56:1, simplified. This ratio produced the highest temperature of 39.0°C and the greatest temperature change of 17.0°C.
Discussion of Theory: The coefficients of compounds in a balanced chemical equation represent the mole to mole ratio of the compounds in a chemical reaction. The ratio of the coefficients is known as the stoichiometric ratio and is the optimum ratio of the compounds to where the most reactants are used up and the most products are produced. When the products of a chemical reaction are unknown, it is not possible
to balance the equation. Therefore, experimental methods must be used to determine the stoichiometric ratio of the compounds. The method of continuous variations was used to determine the stoichiometric ratio between NaClO and N2S2O3 in this experiment. As part of this method, different ratios of the compounds NaClO and N2S2O3were mixed together. An exothermic reaction occurs between these two compounds, and this results in heat being released. By measuring the temperature of each mixture with the different ratios of the compounds, the optimum ratio was found. In this case, the mixture that released the most heat, which meant the most reactants were used up and the most products were produced, contains the optimum ratio of the two compounds. This optimum ratio is the stoichiometric ratio of the moles of the compounds.
Analysis Questions:
1. The differing amounts of NaClO and N2S2O3 combined to form mixtures served as the independent variable. In order for an experiment to yield true results, there can only be one independent variable, one thing that is changing. The volume served as a constant variable. The volume of the reactants needed to be kept constant because the ratios need to be made with the same volume of reactants. If volume was not kept constant, it would have been difficult to decide if the differing volumes were the result of a certain temperature reading or the ratio of the two compounds were the result of a certain temperature reading.
2. The limiting reagent is another way of saying the limiting reactant. The limiting reagent is the reactant in a chemical reaction that is completely used up and limits the amount of product that can be produced.
3. Volume limits the precision of the data in this experiment. Precision is how close a set of measurements are to each other. When different
ratios of NaClO and N2S2O3are mixed and the volume is kept constant, each ratio is going to produce a different amount of heat. There cannot be precision between different ratios of elements even if they have similar temperatures because they are different elements. In order for measurements to have precision, the measurements have to be of one ratio. If the volume were not kept constant, a ratio of 1:21 and 2:42 would yield similar temperature results because the first is the simplification of the second. Therefore, these measurements would have a high precision.
4. The limiting reagent along the upward sloping line of the graph is NaClO. The limiting reagent along the downward sloping line of the graph is the Na2S2O3.
5. Depending on the experiment, the method of continuous variations could use the mass of a precipitate formed, the volume of a gas released, or the changes in color intensity of a reactant or product.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio rather than the ratio associated with the greatest
temperature change because not all of the ratios of the compounds were tested. Although the highest temperature change resulted from the ratio of 40.0 mL NaClO and 10 mL Na2S2O3, a higher ratio could have resulted from a slightly higher or lower ratio. Since these were not tested and measured, using the line of best fit intersection gives a more accurate estimation of the ratio that yielded the highest temperature change.